When I started my research journey, I didn’t expect to spend years thinking about fat.
Like many people, I once thought of fat as just … excess. A silent, expanding organ we often associate with shame, diets, and disease. The deeper I dug into the science, however, the more I realized: Fat is alive. It’s dynamic. It communicates. It adapts. And in the context of obesity, it becomes something else entirely—a cellular battlefield where the rules of regeneration, repair, and even cancer growth rewrite themselves.
Today, I’m a PhD researcher at the University of Utah, where I study how obesity transforms fat stem cells and how these changes expose hidden weaknesses in diseases like diabetes and cancer.
This summary outlines my science— and explains why it matters more than ever.
Adipose tissue—commonly known as fat—lies at the center of my research. Far from being inert, fat represents a highly active organ that plays key roles in energy regulation, immune response, and hormone production.
Stem cells hidden within fat tissue fascinate me the most. These cells have the remarkable ability to become new fat cells, support tissue expansion, and maintain metabolic balance. In the context of obesity, however, these stem cells begin to behave abnormally. They misinterpret cues, trigger inflammation, and, in some cases, fuel disease.
So, what changes in obesity? The answer lies in a tiny, often-overlooked structure: the primary cilium.
The Primary Cilium: A Cellular Compass Gone Awry
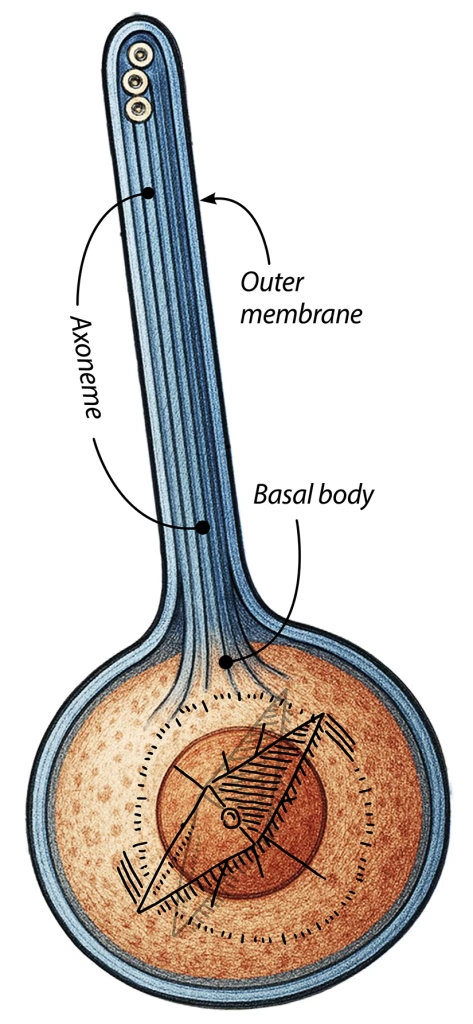
Every fat stem cell includes a primary cilium—a thin, hair-like projection that acts like a molecular antenna. It receives and integrates signals from the cell’s environment and helps decide what the cell should do: divide, differentiate, remain quiescent, or die.
In healthy fat, the cilium functions like a well-tuned satellite dish. Unfortunately, in obesity, the signals become scrambled. The cellular environment becomes flooded with inflammation, mechanical stress, and nutrient overload. The primary cilium can’t keep up, which results in a cascade of miscommunication that pushes stem cells into unhealthy, dysfunctional states.
My research investigates how obesity transforms ciliary signaling pathways, and how these changes affect the fate of stem cells, metabolic health, and even tumor vulnerability.
Cancer Meets Obesity: Uncovering Synthetic Lethal Vulnerabilities
One of the most exciting aspects of my work involves discovering synthetic lethal vulnerabilities in sarcoma, a type of cancer that can arise from fat stem cells.
In simple terms, synthetic lethality occurs when two cellular defects—harmless on their own—become lethal when they combine. Obesity-driven changes to fat stem cells may introduce such defects, revealing new therapeutic targets.
By understanding how obesity reshapes the cellular environment, I hope to identify molecular Achilles’s heels—weak points in cancer cells that we can exploit to develop more precise, less toxic treatments.
A Global Perspective: From Malaria to Metabolism
Before diving into obesity research, I spent more than five years working in malaria and environmental toxicology—fields deeply rooted in global health and environmental justice.
Those early experiences shaped how I view biology, not just as a system of molecules and pathways but also as a product of context—social, environmental, and systemic.
Whether studying how toxins affect vulnerable communities or how parasites hijack red blood cells, I learned that I can never view health as isolated. The same proves true for obesity. Rather than merely representing a number on a scale, it involves a complex interplay among genetics, environment, and molecular miscommunication.
This perspective fuels my current work: to approach obesity not with judgment, but with curiosity, compassion, and scientific rigor.
Why This Work Matters
We currently live in an era when obesity rates appear on the rise globally, affecting both high- and low-income communities. Yet, we still only understand the biological impacts of obesity very poorly. My research helps to bridge this gap in knowledge by offering a molecular lens into how fat tissue adapts—and sometimes breaks down—under chronic stress.
By understanding how stem cells respond to obesity, we can:
- Design more effective therapies for obesity-related diseases.
- Identify biomarkers that signal early tissue dysfunction.
- Develop targeted cancer treatments that exploit obesity-driven vulnerabilities.
In short, this science aims to understand how the body’s most misunderstood tissue can either protect or endanger us—depending on the signals it receives. For me, this work is personal, powerful, and just getting started.
 Esther Okafor is a PhD candidate in the Department of Biochemistry at the University of Utah, where she focuses on obesity and metabolic signaling. She holds a BSc and MSc in biochemistry, and her research explores how physiological cues regulate stem cell fate, especially in fat tissue. She aims to uncover mechanisms that could inform future therapeutic strategies. When not diving into her research, she often listens to contemporary or historical podcasts.
Esther Okafor is a PhD candidate in the Department of Biochemistry at the University of Utah, where she focuses on obesity and metabolic signaling. She holds a BSc and MSc in biochemistry, and her research explores how physiological cues regulate stem cell fate, especially in fat tissue. She aims to uncover mechanisms that could inform future therapeutic strategies. When not diving into her research, she often listens to contemporary or historical podcasts.
This article was originally published in AWIS Magazine. Join AWIS to access the full issue of AWIS Magazine and more member benefits.

